Test Complete
- Questions
- Score
- Minutes
| Overall Results | |
|---|---|
| Total Questions |
| Category Results | |
|---|---|
Quick and Dirty Guide to Acid Base Balance
Category: Medical
Topic: Acid-Base Balance
Level: Paramedic
Next Unit: Acidosis and Alkalosis
17 minute read
Acid-Base Balance Made Simple!
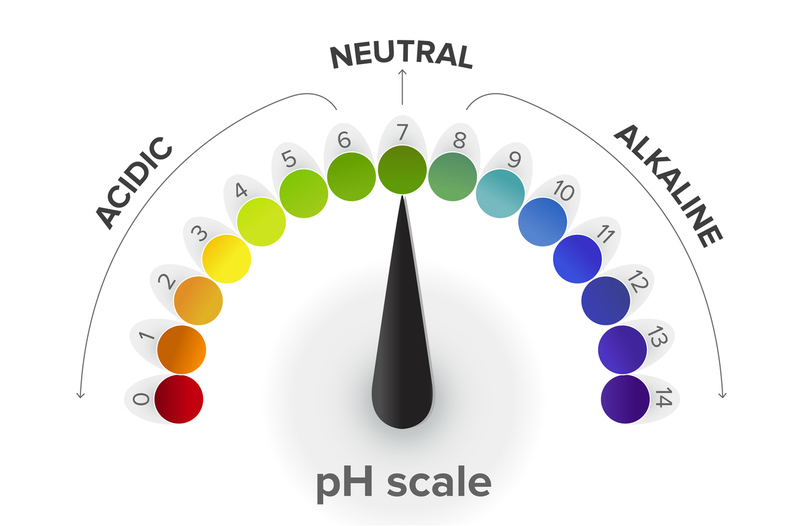
pH is a measurement of acidity. It just measures how acidic something is. When referencing acidity, we're talking about excess hydrogen ions (H+), but we're just going to use the general term "acid." It's a scary foreign phrase, but it just means "percentage of acids." Acids aren't always bad, as in movies. The pH scale goes from 0-14, so neutral is 7 (water is neutral.) The body needs a pH of 7.35-7.45 to maintain homeostasis. 0.5 in either direction away is not conducive to life. So, when we find someone whose acid levels are not conducive to life, we counteract the problem and restore homeostasis. You want to keep the body neither acidic nor alkaline; you want to balance it. This is all Acid-Base balance means.
ACIDIC - TOO MUCH ACID - Lower than 7.35 means too much acid in your system.
NORMAL - Just right! pH between 7.35 and 7.45 is perfect for homeostasis.
ALKALINE - NOT ENOUGH ACID - It just means NOT acidic enough. pH higher than 7.45 is considered Alkaline.
REALLY IMPORTANT
You inhale oxygen; your body turns oxygen into carbon dioxide, and you exhale and remove the carbon dioxide from your body.
Carbon dioxide is "respiratory acid." When you're not breathing adequately, you are not getting rid of this "respiratory acid," and it builds up in the tissues. The extra CO2 molecules combine with water in your body to form carbonic acid, increasing your pH. This isn't good.
How do we measure it?
We can measure the amount of respiratory acid in the arterial blood using blood gases. They measure the amount of each gas in your blood. We measure the pH, the amount of carbon dioxide (PaCO2), and the amount of oxygen in the blood (PaO2).
PaCO2 is the partial pressure of carbon dioxide. We can measure it to see how much respiratory acid (CO2) is in the blood. We use arterial blood gas tests to check it. How much respiratory acid (CO2) should there be? The normal value is 35-45 mmHg (mmHg means millimeters of mercury, a pressure measurement.) The (a) in PaCO2 stands for arterial. If you measure venous blood gasses, the levels are different, and PvCO2 is used. If CO2 is HIGH, it means there is a buildup of respiratory acids because he's not breathing enough CO2 away. If your pH is acidic, and your CO2 is HIGH, it's considered respiratory acidosis. If CO2 is LOW, there are not enough respiratory acids because he's probably hyperventilating too much CO2 away. When pH is high and CO2 is low, this is called Respiratory Alkalosis.
Field Note: On the trucks, pETCO2 (End Tidal Carbon Dioxide) is sometimes used and is expressed as a percentage instead of measuring the pressure in mmHg to measure the effectiveness of ventilation and circulation. A pETCO2 of 5-6% roughly equates to a PaCO2 of 35-45mmHg, so titrate your ventilations to achieve 5-6% pETCo2.
EMT Quick Rule for Testing
Inadequate breathing - Respiratory Acidosis - PH will be low, and CO2 level will be high because he's not breathing it off.
Hyperventilating patients - Respiratory Alkalosis - PH will be high, and CO2 levels will be low because they breathe too much CO2.
Let's make it simple:
If your CO2 is LOW, then DECREASE the rate and depth of ventilations.
If your CO2 is HIGH, then INCREASE the rate and depth of ventilations.
Review
Respiratory Acidosis
NOT Breathing adequately - NOT exhaling enough means you aren't getting rid of CO2; therefore, pCO2 (percent of Co2 is HIGH!)
Clinical Presentation: Altered LOC, Tachycardic, Diaphoresis, headache
Cause: Crappy or no respirations (COPD, Overdose, Pneumonia, Smoke inhalation, Pneumothorax, airway obstruction)
Treatment - Increase rate and depth of ventilations - Bag them so you get rid of some CO2 for them.
Respiratory Alkalosis
Blowing off too much CO2, as in hyperventilation. When there is insufficient CO2 in the blood, the body uses bicarb to compensate for the lack of respiratory acid. (Metabolic compensation)
Clinical Presentation: Numbness or muscle twitch in fingers, toes, seizures
Cause: Shock, DKA Kussmaul Respirations - Deep and FAST breathing! (The body is trying to compensate for its metabolic acidosis by producing a respiratory alkalosis on purpose.) Caused by (anxiety, pain, fever, hypotension, hypoxia, CHF, PE, Sepsis)
Treatment: decrease rate and depth of ventilations. (Calm them down or stop bagging so fast.)
Metabolic
Metabolic means it has to do with metabolism. Metabolism means the chemical changes that sustain life. Oxygen combines with glucose to create energy. Carbon dioxide is a waste product of this process. CO2 is then carried from the blood to the alveoli to the lungs for expiration. We blow off this respiratory acid when ventilations are sufficient. Your body produces a buffer known as alkaline bicarbonate, which binds with excess acids and turns it neutral. Bicarbonate is like Tums for your bloodstream.
HCO3 is the amount of bicarbonate in the bloodstream. HCO3 is just the chemical name for bicarbonate. Normal is 22-28/mEq/l (milliequivalents per liter.) This measures how much Tums are in your blood, binding the excess acid. If your HCO3 (Tums) level is 10, there is not enough of it, so the acid in your blood builds up, and the overall pH of your body goes DOWN! This is called Metabolic Acidosis. To fix it, we increase bicarbonate levels by administering Bicarb via IV! If there's too much, it binds up too much acid, and there isn't enough, so now your pH goes alkaline! This is called Metabolic Alkalosis. Fix it by keeping your patient stable long enough for the bicarb to work itself out.
Metabolic Acidosis
Acid in the blood is used to combine with oxygen and metabolize it. If you do not have enough oxygen-rich blood circulating, the acid is not used and builds up.
Clinical Presentation: Tachycardia, Pulmonary edema, Tachypnea, confusion,n or coma
Cause: Cardiac arrest, renal failure, DKA, diarrhea for a long time, ASA or antifreeze Overdose
Treatment - Increase rate and depth of Ventilations. Consider Bicarbonate - It's like Tums for your blood. It binds up the acid in your body and turns it neutral.
Metabolic Alkalosis
Too much bicarbonate, not enough acid
Clinical Presentation: Seizures, Headache, Dysrhythmias
Cause: Diuretics, cystic fibrosis, chewing tobacco, penicillin, overzealous bicarb administration
Treatment: Keep them alive long enough for their body to use up the excess bicarb.
Handy Dandy Quick Reference

Kidneys
The kidneys also have a fail-safe switch on them. If your pH is too high when it hits the kidney filter, your kidney senses it and flushes MORE acid out with your urine. Similarly, if your kidneys sense that you don't have enough acid, they kick out less acid into your urine. Unfortunately, the kidneys take a while to process it, so it takes hours or days to compensate.
Field Note: Your dialysis patients are chronically acidotic because their kidneys are not participating in the compensation system. So, if your dialysis patient crashes, assuring adequate ventilation and bicarb administration is even more vital.