Test Complete
- Questions
- Score
- Minutes
| Overall Results | |
|---|---|
| Total Questions |
| Category Results | |
|---|---|
INFLAMMATORY RESPONSE - PATHOPHYSIOLOGY
Category: Medical
Topic: Immunology
Level: Paramedic
Next Unit: Advanced Inflammatory Physiology
22 minute read
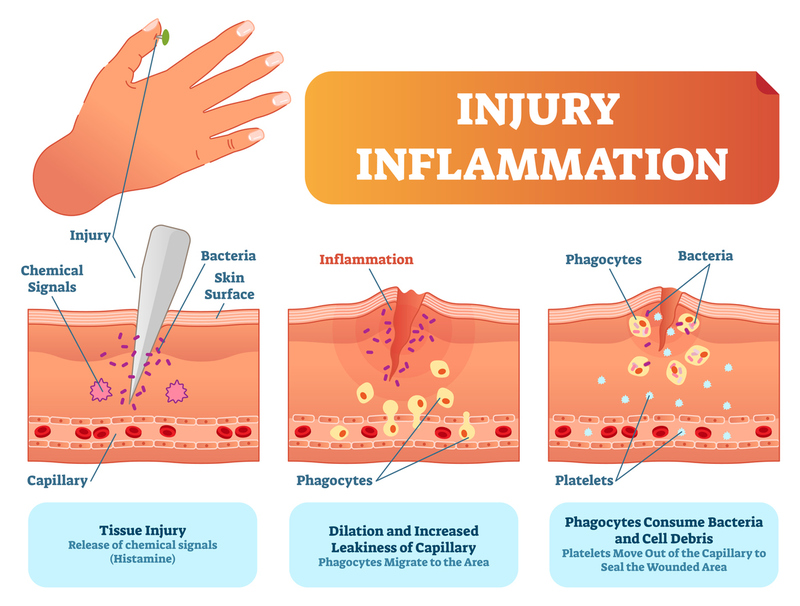
During inflammation, the vasculature of the body responds in three main ways:
- changes in vessel size (and consequently, flow),
- increased vascular permeability, and
- formation of the fluid exudate.
Vessel Changes During the Inflammatory Response
The vessels dilate (slowing the flow of blood) and, with the increased permeability, facilitates movement of white blood cells, as an exudate, out of the vessel to the site where the damage or antigen is.
Extravasation: the process by which certain leukocytes (white blood cell) move to the site of injury. It has 2 distinct steps:
- Leukocyte margination and endothelial adhesion: white blood cells move from the inner portion of the vessel to the outer portion or walls of the vessels. Cytokines released by macrophages bond to the endothelial wall of the vessel and cause leukocytes to “roll” along the endothelial surface. Integrin ligands react further, slowing the leukocytes where they wait to be activated or released.
- Transmigration via the process of diapedesis: causes the adhered leukocytes to move between endothelial cells, which retract and allow the leukocytes to pass through the basement membrane into the surrounding tissue.
Chemotaxis: the process that causes the leukocytes to follow chemical lures towards the source of the inflammation. Once white blood cells reach the site of injury, chemical changes occur and phagocytosis begins.
Degranulation of (and Other Mediators from) Mast Cells
MAST CELLS (WBCs that are part of the immune and neuroimmune systems) contain many granules of histamine and heparin. When activated, mast cells release a variety of vasoactive mediators from cytoplasmic granules and membrane lipids within minutes and also generate and release cytokines and chemokines over hours that recruit other inflammatory cells into the involved tissues.
These mediators are responsible for many of the signs and symptoms of allergic reactions, including flushing, pruritus, urticaria, angioedema, bronchoconstriction, increased vascular permeability, and anaphylaxis.
Inflammatory mediators from mast cells come from two sources,
- cytoplasmic granules and
- membrane lipids:
CYTOPLASMIC GRANULES.
Degranulation: occurs after physical injury damages cells causing chemical changes and IgE binding. (Other important stimulants for degranulation include IgG, complement, and cytokines.)
Degranulation releases pro-inflammatory mediators from granules in mast cells and basophils, specifically,
- Histamine
Causes increased permeability of veins, bronchial and intestinal smooth muscle contraction, increased nasal mucus production, widened pulse pressure, increased heart rate and cardiac output, flushing, T-cell, neutrophil, and eosinophil chemotaxis, increased gastric acid secretion, and airway mucus production.
- Serotonin
Both histamine and serotonin are vasoactive amines that are released during degranulation and cause itching and increases in dilation and permeability of vessels.
- Proteoglycans (heparin)
- Tryptases and other proteases.
Cause anticoagulant activity via inactivation of fibrinogen and inhibition of fibrinogenesis; inactivation of bronchodilators; chemotaxis for eosinophils.
Chemotactic factors released during degranulation attract eosinophils (responsible for combating parasites) and neutrophils (which ingest microorganisms and then release enzymes to kill that type of organism.)
MEMBRANE LIPIDS.
Prostaglandins:
Prostaglandins are synthesized from the arachidonic acid from membrane phospholipids. In contrast to the preformed mediators in the granules, which are released immediately by exocytosis, membrane-derived lipid mediators appear more slowly, generally from 15 minutes to hours after activation. They act upon platelets, endothelium, uterine, and mast cells and regulate inflammation as well as cause constriction or dilation of vascular smooth muscle.
Wheal-and-flare response due to vasodilation and increased vasopermeability are responses to prostaglandins; also, smooth muscle bronchoconstriction, inhibition of platelet aggregation, chemotactic stimulus for neutrophils, and activation of eosinophils.
Leukotrienes:
Leukotrienes increase microvascular permeability and are potent inducers of long-lasting wheal-and-flare responses. They produce bronchoconstriction via prolonged, slow contractions of smooth muscle.
Platelet-Activating Factor:
Platelet-activating Factor attracts eosinophils, neutrophils, monocytes, and macrophages, and it stimulates macrophage cytokine production. It causes bronchoconstriction and vasopermeability.
Plasma Protein Systems
The plasma protein systems are the
- complement,
- kinin, and
- clotting systems.
COMPLEMENT: small proteins found in the blood, synthesized by the liver and normally circulating as inactive precursors in the body. When stimulated by the classical complement pathway (depending on pathogen-binding antibodies), or the alternative complement pathway (does not rely on pathogen-binding antibodies), the complement system activates phagocytes and induces further damage and inflammation to the endothelium—causing coagulation to become uncontrolled, resulting in the formation of microvascular thrombi and other tissue ischemia.
BRADYKININ: released via the kallikrein/kinin system. It is a potent vasodilator that contributes to low systemic vascular resistance (SVR).
CLOTTING SYSTEM: While inflammation initiates the clotting system, it also decreases the activity of natural anticoagulant mechanisms and slows the fibrinolytic system. As such, inflammation is pro-coagulation.
Clotting takes two paths:
- the cellular aspect (platelets), and
- the protein (coagulation factor) aspect.
The leaking of blood through the endothelium initiates changes in the platelets and exposes the tissue to plasma Factor VII, which forms fibrin.
Cellular Aspect: Primary hemostasis is the process where platelet changes include localization to the extracellular matrix and vasoconstriction. Here, platelets are activated by different proteins, causing the platelets to adhere to the site of the injury and release contents of stored granules in the blood plasma. The contents of the granules, in turn, activate more platelets. The platelets then change shape from spherical to stellate and bind fibrinogen, which aids in platelet aggregation.
Protein Aspect: Secondary hemostasis begins and consists of two pathways.
The intrinsic pathway—contact activation pathway, caused by trauma inside the vascular system—is less important for coagulation and plays a larger role in inflammation. It begins with a reaction between collagen and high-molecular-weight kininogen, prekallikrein, and other proteins. (This process is slower than the extrinsic pathway.)
The extrinsic pathway—tissue factor pathway: External trauma generates a “thrombin burst” by which thrombin (most important factor in coagulation cascade) is released very rapidly. Much faster than the intrinsic system, it meets with the intrinsic pathway to result in clot formation in what’s called the common pathway.
The plasma protein systems all work together to limit and control the inflammatory process, prevent and limit infection or further damage, interact with the adaptive immune system, and prepare the injured site for healing. The system has built-in antagonists and receptors to keep levels of histamine under control and to know when production of components needs to cease.
Lymphokines (Cytokines from Lymphocytes)
LYMPHOKINES are a type of cytokine produced by lymphocytes that direct the immune system response by signaling between its cells.
Lymphokines activate macrophages, transform lymphocytes, and mediate cell-mediated immunity. Lymphocytes detect very small concentrations of lymphokine and then concentrate where the immune response is needed.
Regulation is via migration-inhibitory factor and macrophage-activation factor.
Migration-inhibitory factor is a key regulatory cytokine that promotes and regulates cytokine secretion and helps to regulate the immune response.
Macrophage-activation factor determines how a macrophage will respond: classically activated, wound healing, or regulatory. Regulatory macrophages are produced as a product of macrophages and glucocorticoids.
Interferons
INTERFERONS: Interferons are a group of signaling proteins (cytokines) made and released by host cells in response to the presence of several pathogens. They interfere with viral replication to protect cells during a virus infection. The also activate immune cells and increase host defenses by up-regulating antigen presentation.
Interferon type I is produced by fibroblasts and monocytes when the body recognizes a virus has invaded it.
Interferon type II, known as the "immune interferon," is released by T-helper cells type 1 (Th1) and inhibits proliferation of (Th2). Types I and II participate in regulating and activating the immune response.
Systemic Response to Acute Inflammation
Local inflammatory responses include
- vascular dilation and
- increased capillary permeability, as well as
- exudation that fills wounds with pus (composed of serum, fibrin, and white blood cells).
The systemic response of acute inflammation includes:
- fever (activated by endogenous or exogenous pyrogens that assist the healing process),
- leukocytosis (activated by the increased release of leukocytes from bone marrow, decreased margination of leukocytes onto vessel walls, decreased extravasation of leukocytes from the vessels into tissues, or an increase in a number of precursor cells in the marrow),
- increase in circulating plasma proteins or acute-phase reactants (released by the liver in response to cytokines in the bloodstream) that allows for opsonization, recruitment, coagulation, and other roles.)
Systemic Response to Chronic Inflammation
Chronic inflammatory responses are caused by an unsuccessful acute inflammatory response and/or persistent infections or antigens.
Characteristics of a chronic inflammation response are long-term infection, neutrophil degranulation and death, lymphocyte activation, fibroblast activation, infiltration (pus), and tissue repair or scarring.